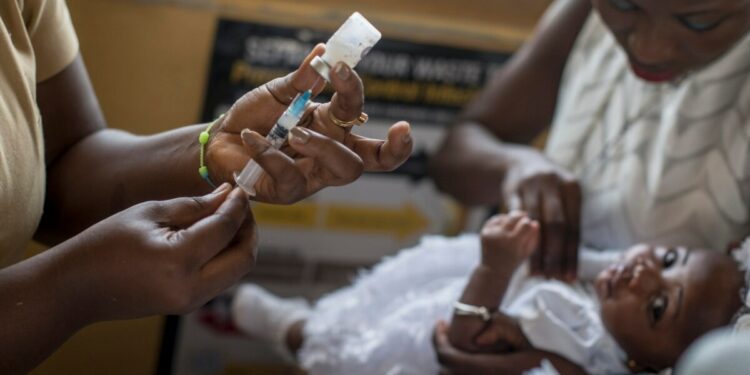
This week, Nigeria’s pharmaceutical regulatory authority has joined Ghana in supporting the new R21 vaccine, making them the first countries globally to do so. This vaccine was developed by Oxford University scientists and manufactured by the Serum Institute of India.
Read More: 5 Tech Hacks for Students to Instantly Improve Productivity
African countries that do not have extensive resources for drug regulation have previously relied on the U.N. agency to initially review new medicines. At present, there is a lack of public access to comprehensive information regarding the malaria vaccine’s outcomes in large-scale trials. Additionally, it remains uncertain how low-income countries will afford the cost of the vaccination.
However, the urgency of addressing a disease that kills more than 600,000 people annually, most of them children under age 5 in Africa south of the Sahara, and recent efforts to enhance drug oversight in the region, are changing the process.
The World Health Organization stated in a high-level meeting this week that regulatory authorities from at least 10 African countries are currently examining trial data to evaluate the vaccine. It is anticipated that additional countries will grant approval for the vaccine in the next few weeks.
“We expect many more countries to come through,” Mary Hamel, the WHO’s malaria vaccine implementation head, told the expert meeting on Tuesday. “They are sovereign countries that can make their own decisions for their vaccines.”
Read More:Giving the Most Vulnerable their Dignity Back
The countries that could be affected were not specifically identified by her. However, it is worth noting that Tanzania and Kenya have stringent regulatory bodies and comparatively high prevalence rates of the illness.